The water treatment industry has developed and evolved over the years to continue to find new ways to produce degassed water,
with many advances in both the technological methods of treatment as well as the refinement of the existing methods. The evolution of water treatment has been driven by the need for increased demand and over-safety standards.

All human cultures on our planet share a single commonality and that is the dependency on water to survive.
Many existing technologies such as “degasification” have evolved with higher efficiency to meet the demand changes and provide safety to consumers and to systems. Degasification refers to the removal of dissolved gases from liquids and the science to degasify water is based upon “Henry’s Law” or to be exact the “proportionality factor is called Henry’s law constant” and was developed by William Henry in the early 19th century.
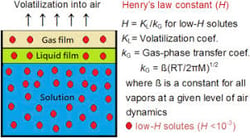
Henry’s Law states that the amount of dissolved gas is proportional to its partial pressure in the gas. The most effective method to perform degasification is with the packed vertical tower called a degasifier or decarbonator. When water enters at the top of the tower gravity feeds downward across a media bed. The media bed acts to reshape the water over and over again exposing any dissolved gas molecules to the surface of the water droplet. At the same time that the water is traveling down the interior of the tower an air flow is introduced in a cross-current method either by force or by induction that passes over the water droplets and “strips” the gas molecules out of the water. The gases that are stripped then leave the tower through the exhaust at the top of the tower. This is the “basics of water degasification”.
There are many reasons to perform degasification
and not all are related to water treatment. There are forms of degasification needed during the production of chemicals, medicines, and even cosmetics but the most prevalent need is water. When water is utilized to produce steam the dissolved gases like CO2 (carbon dioxide) must be removed prior to the water entering the boiler to guard against and prevent corrosion and scaling. When CO2 is left in the water it can form carbonic acids which cause corrosion and reduces the performance and shortens the life of the equipment needed to produce the steam. When water is being treated for consumption it is also often degasified to remove CO2 and other dissolved gases such as Hydrogen Sulfide (H2S) or as commonly referred to by consumers “Rotten egg gas”.
 Degasification is the most cost-effective
Degasification is the most cost-effective
method to treat water to remove carbon dioxide (CO2) which will elevate the water's pH by stripping out the carbon dioxide (CO2) gases. Often the pH of water is first lowered prior to reverse osmosis to help prevent membrane scaling so after the water leaves the membrane filtration process the pH may still be in the acidic range. If degasification was not utilized to lower the pH then more chemicals would be needed such as caustic (a base) to raise the pH back up to a neutral level of 7. The cost of degasification with a degasification tower is far more cost-effective than chemical use when you review the operating cost of a typical water treatment facility. At DeLoach Industries a 60-year-old designer and manufacturer of degasification towers they have continually advanced their systems to improve efficiency and lower operating cost. Items like the distribution system is a key component to begin the degasification process when the water first enters the treatment tower. The type of media utilized and the air vane flow system are also key items that impact the performance of a degasification tower. DeLoach Industries is currently designing one of the first “smart” degasification towers on the market which will be released in 2018 to better serve the water professional. For more information or to learn more contact the professionals at DeLoach Industries Inc. at (941) 371-4995.
Receive our free Hydraulic Data E-Book





