Water demineralization is also called deionization and is a process known as “Ion Exchange.”
In simple terms, water demineralization is “Water Purification.” The process involves removing dissolved ionic mineral solids from a feed-water process, typically for “Industrial” water applications. Still, it can also be utilized to remove dissolved solids from a water process for “Aquaculture,” “Food and Beverage,” and the “Municipal” markets.
Why is demineralization utilized? It can remove dissolved solids to near distilled water quality at a much lower capital and operational cost than other treatment processes such as membrane softening (Reverse Osmosis). Demineralization applies the science known as “Ion Exchange,” which attracts negative and positive charged ions and allows either to attach themselves to a negative ion depending on their respective current negative or positive charge during what is known as a resin cycle. In other technical articles, we will explore and go into more specific details on the science of the ion exchange process. Water that has dissolved salts and minerals has ions, either negatively charged ions known as “Anions” or positively charged ions known as “Cations.” To treat the water and remove these contaminants, the ions in the water are attracted to counter-ions, which have a negative charge. In a demineralization treatment process, there are pressure vessels that hold resin beads which are typically made of plastic. The beads are made from a plastic material with an ionic functional group that allows them to hold and maintain an electrostatic electrical charge. Some of these resin groups are negatively charged, referred to as “Anion” resins, while others hold a positive charge and are called “Cations” resins.
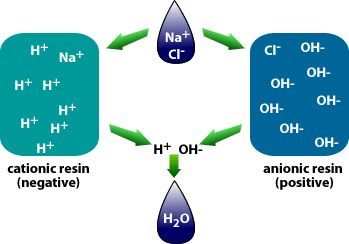
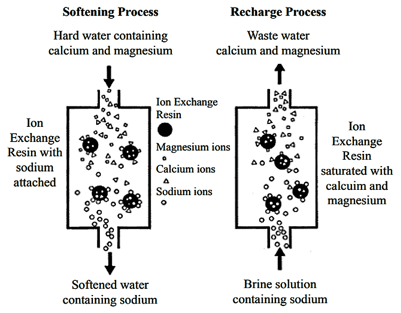
There are different applications to apply Ion exchange technologies, which is why you will often hear different terminology interchanged like deionization and demineralization. The raw water quality and the specific application will dictate the type of ion exchange process needed. For example, if the water contains a high level of hardness, the water will most likely contain Ca2+ or Mg2+ dissolved solids possessing a positive charge. To replace these hard ions, it is typical to utilize a resin bed with a salt ion like Na+. As the water passes over the resin bead material within the pressure vessel. The hard ions are replaced with the salt ion; therefore, all the hardness within the water is removed. However, the water will now contain a higher concentration of sodium ions, and this must be considered during the evaluation and selection process of the type of resin material to utilize for the specific application. If the water application requires high purity and the removal of as many solids as possible, then the term or process selected is referred to as demineralization.
In these applications, the dissolved solid ions are removed utilizing hydrogen ions H+ and hydroxyl ions OH-. The net yield of this process is H2O or water. So it would be simpler to say that demineralization is a higher form or more target-specific form of ion exchange in that it removes almost all solids near distilled water why do other forms of Ion-exchange remove certain types of dissolved minerals but may replace one for another? This may resolve a hardness or scaling problem but allow an increased level of other forms of dissolved solids, such as sodium. DeLoach Industries Inc. is one of the oldest water treatment equipment manufacturers in the United States with its 63-year history. At DeLoach Industries Inc., after their experts assist the customer or their engineer with reviewing the water analysis and the specific application, they select the type of equipment, size, and specific resin beds needed to achieve the desired water quality by the customer. In addition, the engineering design team at DeLoach Industries applies years of experience in treatment plant operations and the need to reduce operating costs in every design. The design team will identify critical components that will reduce operational costs and potential headaches for their customers and make design suggestions backed by written engineering calculations showing operational cost reductions. The addition of a decarbonator to a process stream is an example of a recommendation by DeLoach Industries to remove carbon dioxide to extend the life of a resin bed and reduce regeneration cycles.
Demineralization systems consist of a minimum of at least one pressure vessel. Still, they often require two for industrial applications. The DeLoach Industries systems are skid mounted at the factory, including chemical feed storage tanks, regenerative automatic dosing system, and PLC fully automated control system that is factory installed pre-wired. In addition, all plumbing and valves can be manual or automated depending upon the customer's selection and are included, factory-installed, and tested on every DeLoach Industries system. All the DeLoach Industries systems are factory tested, include the resin bed charge, and come with full support, including setting up, start-up, balancing and testing, and training. Demineralization is utilized in multiple market industries and is widespread in the industrial markets to treat boiler feed water. It is not uncommon to see both membrane treatment and demineralization utilized in series to create higher-purity water.
Almost always, in a demineralization process, you will want to incorporate a “Decarbonator” or Degasification” process to remove all CO2 (Carbon Dioxide). By removing the CO2, you will extend the life of the resin bed within the demineralization process. If you need to remove hardness, calcium, or other dissolved solids from your water, please contact the experts at DeLoach Industries. They will assist you on your next project and choose the design selection for your specific needs. If your existing system has operational problems, the engineering support team at DeLoach Industries will be happy to help you solve your problem. Feel free to contact DeLoach Industries at 941-371-4995 or look us up at DeLoachIndustries.com so they can be replaced during the process.
As an example: