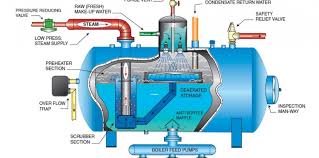
In the United States manufacturing industry, an astonishing 400 million gallons of water per day (MGD) is consumed to generate steam.
Out of this amount, approximately 60 MGD is sent to blow-down drains, while another 300 MGD is used for direct injection of steam. The common denominator in all of these processes is the need for purified and treated water. Without proper treatment, manufacturers would face frequent shutdowns and increased capital expenditure, significantly impacting their cost of goods. One effective method of water treatment to protect boilers is through degasification and deaeration.
Degasification towers play a crucial role in removing harmful gases such as hydrogen sulfide (H2S), carbon dioxide (CO2), and often dissolved oxygen (DO). The elimination of these corrosive gases is vital for enhancing the lifespan and efficiency of boiler systems. If these gases are allowed to remain in the boiler feed water, particularly carbon dioxide (CO2), it can lead to disastrous consequences, including higher operating costs and reduced system longevity. Carbon dioxide (CO2) can convert into carbonic acid, creating a corrosive environment for the boiler and other critical components. In cases where an ion exchange process is implemented prior to the boiler, the presence of carbon dioxide (CO2) can drastically increase regeneration costs as the resins are consumed. By removing carbon dioxide (CO2), the life of the resin is extended, and the pH of the water is elevated, reducing the need for additional chemicals and further lowering operating costs.
Read More
Topics:
water treatment issues,
degasification,
iron oxidation,
water treatment,
water distribution system,
advanced treatment solutions,
water plant,
hydrogen sulfide (H2S),
Decarbonation,
ION Exchange Resin,
feed water,
De-Aeration,
steam generation,
steam generating boilers,
carbon dioxide,
steam,
decarbonator,
boiler system,
degasifier,
gases,
RO membrane,
carbonic acid,
RO system,
H2S Degasifier,
Boiler feed water
Avoid problems with calcium chlorite and corrosive gasses with your odor control scrubber.
When planning or designing an odor control system, one should pay close attention to several key variables that can cause havoc on a chemical odor control scrubber when trying to treat hydrogen sulfide or ammonia gases. The need for odor control occurs in many different forms. It is essential to understand the process that is creating the odorous or corrosive gas and the need for odor control & air emissions treatment.
First, begin to identify
all the potential obstacles that may creep up later after the chemical odor or corrosive gas control system goes online, like acid or caustic consumption. For example, chemical odor control systems designed for water treatment for the municipal industry are typically needed and attached to a degasification or decarbonation process, often needed to treat hydrogen sulfide (H2S). However, designers often may not pay close enough attention to the type of water process available for “make-up” water for the chemical scrubber. The addition of caustic can create scaling or fouling. This unknown variable of the makeup water quality can lead to a complete tower shutdown if the chemical scrubber distribution and media bed scales or fouls. The most commonly used chemicals for a hydrogen sulfide (H2S) scrubber are either chlorine in the form of sodium hypochlorite or caustic in the form of caustic soda. Both of these chemicals are common to a water treatment facility and are already in place to adjust and control pH.
The makeup water plays a significant role in the operation of a chemical scrubber.
When water containing high hardness levels is used as the source for the makeup water, your chemical scrubber can become fouled, and scaling can occur in a matter of hours, depending on the alkalinity and salts within the water. Solidification can occur from the scaling when combining sodium hypochlorite and raw feed water at specific pH ranges and these ranges are usually the range needed to achieve peak performance. Calcium chloride will form, and your chemical odor control scrubber will become a solid chunk of calcium chlorite making, making the ability for water or air to pass freely through the media packing next to impossible. No matter what type of media packing is utilized in the odor control or gas scrubber, it can foul and scale if the water chemistry is incorrect. Trust me when I say “been there and done that”! I have seen operators who have allowed a chemical scrubber to become out of balance with pH control and completely solidify the tower column to the degree that neither air nor water passage is possible. The problem can still occur with ammonia scrubbers but are different with different sets of parameters.
Read More
Topics:
odor control,
water treatment,
advanced treatment solutions,
biological scrubber,
water plant,
odor control scrubber,
hydrogen sulfide (H2S),
calcium carbonate,
media packing,
pH levels,
Alkalinity,
Langilier index (LSI),
scaling,
chlorine,
caustic,
ION Exchange Resin,
Safe drinking water,
dissolved gases,
De-Aeration,
carbon dioxide,
oxygen,
degasifier,
gases,
H2S Degasifier,
calcium chlorite
The water treatment industry has developed and evolved over the years to continue to find new ways to produce degassed water,
Read More
Topics:
water quality,
degasification,
pH levels of water,
water treatment,
advanced treatment solutions,
water plant,
safety,
hydrogen sulfide (H2S),
Chemical Odor,
media packing,
pH levels,
Decarbonation,
dissolved gases,
wastewater,
Global,
carbon dioxide,
decarbonator,
degasifier,
gases,
RO membrane,
H2S Degasifier,
degassed water
Protecting Your Pharmaceutical Water: Ensuring Quality and Efficiency in Water Treatment
In the pharmaceutical industry, the removal of dissolved gases from water is a critical step in the water treatment process. However, it is essential to select the appropriate method of removing these gases, as the wrong choice can have detrimental effects on vital process water equipment such as steam boilers and distillation columns. Failure to address high levels of carbon dioxide (CO2) in the water can lead to the formation of carbonic acid, which corrodes and damages both the steam boiler tubes and distillation columns. To mitigate these risks, the implementation of a degasification tower or "Degasifier" is crucial, as it effectively removes dissolved gases like hydrogen sulfide (H2S) and carbon dioxide (CO2) to acceptable levels below 7 parts per billion (ppb).
Utilizing a degasification tower offers a cost-effective solution to reduce and eliminate gases in the water stream. In comparison, alternative methods such as reverse osmosis (RO) membranes require additional steps, including pH adjustment, to achieve similar results. The conversion of carbon dioxide (CO2) into carbonates can result in increased membrane fouling and elevated capital costs for the RO system. By implementing a degasification system, businesses can achieve optimal performance, minimize membrane fouling, and benefit from cost savings in both capital and operational expenses.
Read More
Topics:
degasification,
water treatment,
hydrogen sulfide (H2S),
dissolved gases,
pharmaceutical water,
carbon dioxide,
degasifier,
gases,
RO membrane,
carbonic acid,
RO system
The Term referred to as “Degasification” or "Decarbonation" and how they work
Relates to the process of the removal of suspended gas or solids that are converted to a gas-based upon certain criteria during water filtration, treatment, membrane filtration, or attempting to adjust pH. When removing (CO2) the process is often referred to as “Decarbonation”, when removing (H2S) the process is often referred to as “Degasification”.
Degasification is the most economical method for
the removal of Hydrogen Sulfide (H2S), Carbon Dioxide (CO2), and Oxygen (02) can all be removed by “Degasification”. The other variables are the total inlet water flow rate, the inlet feed temperature of the water, the ambient air temperature, the inlet concentrations that can be expressed as parts per billion (ppb), parts per million (ppm) or Mg/l, and the desired effluent removal levels also expressed in the same method. It is also important to fully understand the actual application and the use of the water to determine how critical maintaining critical levels are and what impact variations will create for the final use. Understanding these variables will aid you in the design of the system and any additional redundant systems needed to assure full compliance with standards.
Read More
Topics:
degasification,
water treatment,
advanced treatment solutions,
hydrogen sulfide (H2S),
pH levels,
Decarbonation
Do you need to remove or increase your reverse osmosis system's hydrogen sulfide removal efficiency?
The industrial water treatment market has many forms of water treatment processes. Most of us would agree that maintaining high water standards and quality requires using multiple treatment systems to achieve results. Let’s face it, we do not win or get a “that a boy” when we design and build the best reverse osmosis system.
When we turn the brand-new water system on, the water has a "rotten egg odor." Yes, that is an embarrassing moment!
The problem is we typically design around what we can see or read. When was the last time you reviewed a water sample that provided details of how much-dissolved gas was in the water? Most likely never. A typical water treatment system may deploy reverse osmosis as the primary treatment method, and why true RO will remove particles that have size and weight (ions and molecules) typically defined as a certain size (micron), but RO does nothing to remove the dissolved gases that are already entrained within the water naturally or were created by adjusting the pH.
Read More
Topics:
water treatment issues,
water quality,
degasification,
pH levels of water,
water treatment,
advanced treatment solutions,
hydrogen sulfide (H2S),
pH levels,
Alkalinity,
Langilier index (LSI),
H2S Degasifier,
H2S H2O,
removing hydrogen sulfide in water
Water treatment towers and storage tanks are high places that require special precautions when entering. While the majority of people who enter these locations for work can be trusted, there are some hazards that make it more important than usual to follow safety procedures.
These locations can get very hot and humid, and can also be filled with harmful chemicals and microorganisms that can cause serious health issues if inhaled or absorbed through the skin. Therefore, the general standard for workplace safety is much higher when entering locations like these.
Make sure you have read and understood the following information about safety when entering a water treatment plant. It will help you understand how to stay safe and protect yourself from harm when entering a water treatment plant. normal installation, maintenance, or even emergency repairs, it is often required to enter into a water treatment tower (degasifier, air stripper, decarbonator, or clear well/ storage tank). When this occurs, full safety protocols should be followed at all times, in accordance with OSHA regulations. A tower or tank B classification is a "Confined Space" location. For more information visit the OSHA confined space regulations page.
In addition, there are other safety risks that an operator or technician can be exposed to while inside these types of closed locations. The risk can come from fumes of hydrogen sulfide (H2S), chlorine from an injection line, or a lack of oxygen O2. A proper confined space permit should be prepared and only technicians with proper training and certifications should enter into these types of confined spaces.
Read More
Topics:
water treatment issues,
water quality,
odor control,
water treatment,
advanced treatment solutions,
biological scrubber,
water plant,
safety,
odor control scrubber,
hydrogen sulfide (H2S),
Chemical Odor,
media packing,
scaling,
caustic,
Safe drinking water,
dissolved gases,
wastewater,
carbon dioxide,
degasifier,
gases,
Ammonia,
what is a scrubber,
Hydrogen Sulfide formula,
Deagasification,
Filter Media,
DeLoach Industries, Inc.,
Drinking Water,
Clean Water,
Contaminated Water,
OSHA

Following NSF/ANSI 61 regulations when designing and selecting the materials for the manufacturing of water treatment equipment.
It is important to understand what regulatory standards or constructions standard may be required to be compliant. This includes the designing and fabrication of systems such as reverse osmosis utilizing membrane technology, decarbonation of Carbon Dioxide, degasification of Hydrogen Sulfide, and water filtration for the removal of micron particles from potable and nonpotable water processes.
One requirement that engineers and manufacturers often encounter is called NSF /ANSI 61. NSF is an international and nonprofit, nongovernmental organization that is focused and dedicated to public health and safety as it relates to potable water systems and their components. NSF/ANSI 61 developed and established minimum requirements for the control of potential adverse human health effects from products and their components that contact with drinking water.
DeLoach Industries Inc. manufactures multiple types of water treatment equipment and adheres to strict compliance with NSF/ANSI 61 standards with all of their manufacturing procedures and practices. This strict adherence assures owners that water treatment equipment like decarbonation and degasification towers, reverse osmosis, and ion exchange that the equipment and material are all in full compliance with the NSF/ANSI 61 requirements.
Read More
Topics:
water quality,
water treatment,
advanced treatment solutions,
About DeLoach Industries,
fabrication,
contact molded process,
hydrogen sulfide (H2S),
Decarbonation,
wastewater,
carbon dioxide,
decarbonator,
H2S Degasifier,
ansi61,
nsf/ansi61