Why Aerator Towers Are the Most Cost-Effective Way to Oxidize Iron
Iron contamination in water systems is one of the most common—and costly—challenges faced by municipalities, industrial facilities, and private utilities. From staining and foul taste to clogged pipes and equipment damage, untreated iron can quickly escalate operational expenses.
Read More
Topics:
aeration,
Aerator,
Industrial water treatment,
aeration tower design,
ferric hydroxide formation,
groundwater iron removal,
Iron oxidation aeration,
oxidizing iron in water,
dissolved oxygen water treatment,
iron removal systems,
water treatment aeration
Aeration, Decarbonation, or Degasification? How to Choose the Right Water Treatment System — and Avoid Costly Mistakes
Read More
Topics:
degasification,
aeration,
Decarbonation,
municipal water systems,
DeLoach Industries,
wastewater treatment systems,
Industrial water treatment,
aeration system,
CO2 removal,
hydrogen sulfide removal,
degasifier tower,
pH control system,
carbon dioxide stripping
Water treatment is the backbone of industrial processes, ensuring that systems run smoothly and efficiently. One of the essential components in this field is the degasification and decarbonation system. By effectively removing gases from water, these systems enhance water quality and minimize corrosion risks. Here we'll cover the intricacies of hydraulic flow optimization for degasification systems and reveal the best practices for avoiding common pitfalls in system design.
Read More
Topics:
degasification,
aeration,
water treatment,
degasifier,
DeLoach Industries, Inc.,
Induced Draft,
Industrial water treatment,
Water Treatment Technologies,
Forced Draft Degasification,
Elevated Degasification Systems
Industrial water treatment is a cornerstone of efficient and sustainable industrial operations. The presence of dissolved gases, particularly carbon dioxide (CO2) and hydrogen sulfide (H2S), can wreak havoc on equipment and processes. Understanding how to effectively manage these gases through degasification is crucial. In this comprehensive guide, we'll explore the mechanics and benefits of forced draft and induced draft degasification towers, empowering you to make informed decisions for your water treatment systems.
Read More
Topics:
degasification,
aeration,
water treatment,
degasifier,
DeLoach Industries, Inc.,
Induced Draft,
Industrial water treatment,
Water Treatment Technologies,
Forced Draft Degasification
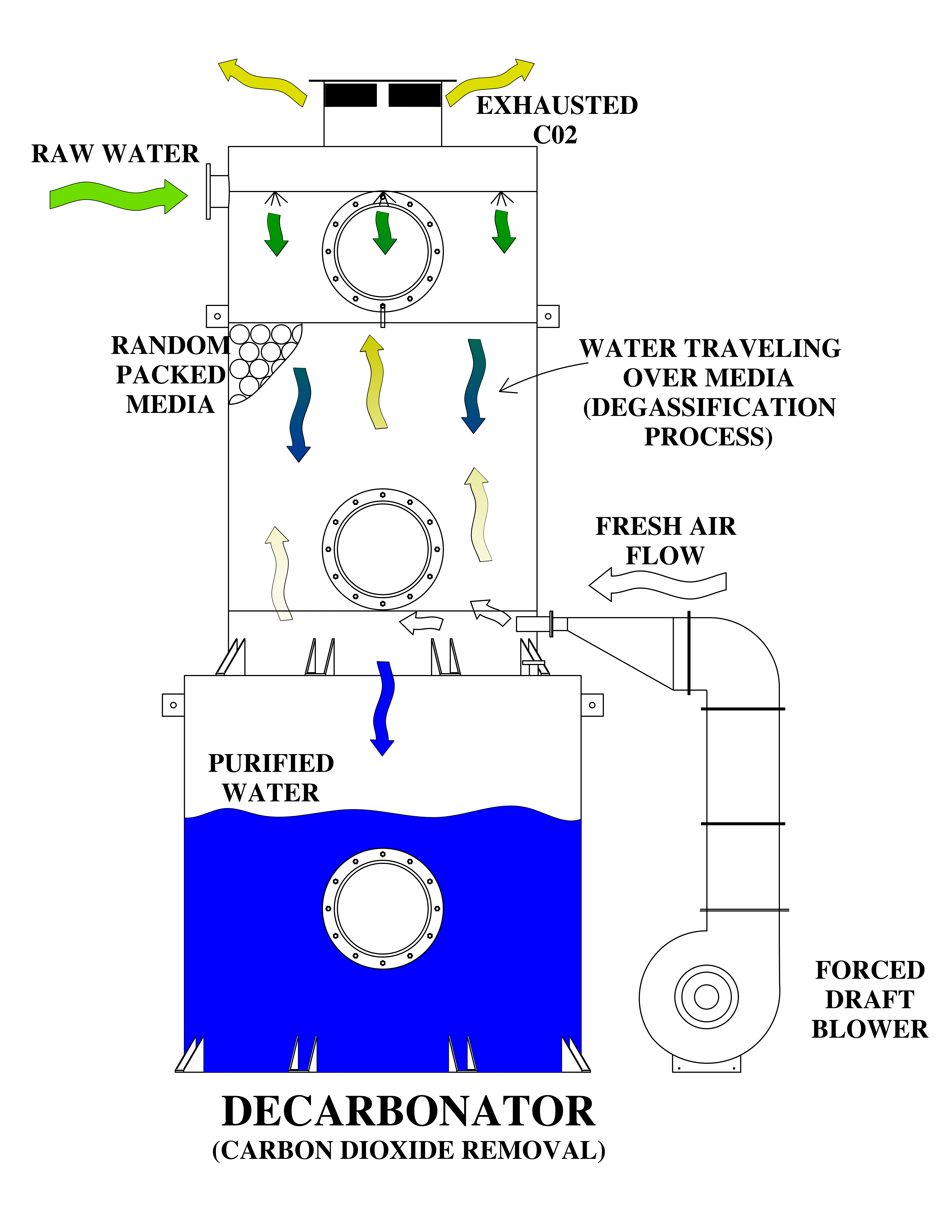
Requires an application commonly referred to as either “Degasification” or "Decarbonation" and it requires the use of a piece of water treatment equipment called either a “degasifier” or a “decarbonator”.
Both of these are similar in nature and are designed for Carbon Dioxide (CO2) removal from the incoming water. A properly designed decarbonator can remove 99.99% of the free carbon dioxide gas that is present in the water stream. One of the primary reasons for utilizing a decarbonator or degasifier for the removal of carbon dioxide gas is the raise the pH of the water without the need to add caustic. resulting in high-purity water.
The other reason is the remove the CO2 prior to treating the water with Ion Exchange which utilizes Anion or Cation resins to reduce the regeneration cycles for the resin beds. High concentrations of CO2 consume the ion charge within the resins and require more frequent regeneration cycles. The difference between anion and cation resins is that one is positively charged (anion) and the other is negatively charged (cation), cation resins, attract positive ions with their negative charge.
The term decarbonation describes the process of the removal of suspended gas or the conversion of carbonic acids into free Carbon Dioxide. Carbonic Acid (H2CO3) is stable at normal ambient anhydrous conditions. However, Carbonic Acid decomposes when not stable and in the presence of any water molecules to form carbon dioxide (CO2). The Carbonic acid breaks down when present in water and it is converted to a gas based upon certain conditions. It is common to have CO2 present in water requiring a decarbonation process when utilizing certain types of water filtration such as membrane filtration with reverse osmosis or it can be present when the need to adjust pH is required. When removing (CO2) the process is often referred to as “Decarbonation”, when removing (H2S) Hydrogen Sulfide the process is often referred to as “Degasification”.
Read More
Topics:
water treatment issues,
degasification,
pH levels of water,
aeration,
iron oxidation,
water treatment,
water plant,
bicarbonate,
hydrogen sulfide (H2S),
pH levels,
Decarbonation,
ION Exchange Resin,
dissolved gases,
De-Aeration,
wastewater,
carbon dioxide,
oxygen,
decarbonator,
degasifier,
gases,
carbonic acid,
H2S Degasifier
The Basics of Water Decarbonation
and the removal of carbon dioxide (CO2). The need to remove (CO2) is essential in most Aquaculture, Municipal, Industrial, and Food & Beverage Processes To understand you must familiarize yourself with Henry’s Law.
Henry's Law defines the method and proportional relationship between the amount of a gas in a solution in relation to the gas's partial pressure in the atmosphere. Often you will see and hear various terms like degasification, decarbonation, aeration, and even air stripping when discussing the removal of dissolved gases and other convertible elements from water. Understanding the impacts that Carbon Dioxide (CO2) can have on both equipment and aquatic life provides the basic reasons why the need to decarbonate water, exists. Carbon Dioxide (CO2) can exist naturally in the raw water supply or be the result of ph control and balance. In either case, the process called Decarbonation or Degasification provides the most cost-effective and efficient manner to reduce or tally remove (CO2) from the water. In addition to Carbon Dioxide (CO2), water can contain a variety of other contaminants that may impact the removal efficiency of the Carbon Dioxide. A variety of elements as well as dissolved gases such as oxygen, nitrogen, and carbon dioxide (CO2). A full analytical review of the water chemistry is required to properly design and size the “Water Treatment” process.
Breaking the bonds in water releases a dissolved gas
such as carbon dioxide (CO2) you must change the conditions of the vapor pressure surrounding the gas and allow the gas to be removed. There are many variables to consider when designing or calculating the “means and methods” of the removal of carbon dioxide (CO2). When I refer to the means and methods. I am referring to the design of a decarbonator and its components. The means equals the size and type (Hydraulic load) of the decarbonator and the “method” equals the additional variables such as the cubic foot of airflow (CFM) and “Ratio” of the air to water to accomplish the proportional condition needed to remove the carbon dioxide (CO2).
Read More
Topics:
water treatment issues,
degasification,
pH levels of water,
aeration,
iron oxidation,
water treatment,
water plant,
bicarbonate,
hydrogen sulfide (H2S),
pH levels,
Decarbonation,
ION Exchange Resin,
dissolved gases,
De-Aeration,
wastewater,
carbon dioxide,
oxygen,
degasifier,
gases,
carbonic acid,
H2S Degasifier,
removal of CO2 from water
Converting Ferrous (Fe+2) (soluble) iron to Ferric (Fe+3) (Particulate/Solid form).
The iron must first be exposed to air or an oxidizing agent. Aeration System is the most cost-effective method to oxidize ferrous iron for its removal from water. In many areas around the globe, municipal and industrial operations need to remove naturally occurring iron (Fe) from the water to prevent damage to other equipment and improve water quality. Removing iron from the water must first be oxidized using the most widely accepted and cost-effective method called aeration. The aeration process changes the iron from its Ferrous (Fe+2) state (soluble) to ferric (Fe+3) colloidal participation. Did you know that Iron occurs naturally and is found in the earth’s crust? It occurs in both groundwater as well as surface waters and is not known to cause any harmful effects on humans or animals.
Iron does cause problems, though, for municipal facilities and their customers by impacting laundry operations and causing stains on buildings and on plumbing fixtures. Iron also promotes and facilitates the growth of iron bacteria in water, creating a problem for distribution lines and piping systems. Once the lines become blocked, this impacts the ability to distribute water to the customer. Iron bacteria also become detectible even at low concentrations and impact the taste of the water. The U.S. Public Health Service Drinking Water Standard set a recommended maximum level of 0.3 mg/L in public water supplies.
In industrial applications, iron can cause server damage in boiler systems.
Cation and Anion systems, piping and nozzles, and other equipment. In addition, for other industries such as food and beverage, brewery, semiconductor, or the production of chemical products, iron can interfere with the manufacturing or brewing and canning process, lowering the quality of the final product. In groundwater and anaerobic surface water, iron is normally present in its soluble form called “Ferrous” iron Fe+2 when the pH is in a certain range. When the same water is in contact with air, that allows the air to diffuse into the water, or when any form of an oxidizing chemical is added to the water, the iron is converted into its oxidized form called “Ferric” Fe+3, becoming a solid. The ferric iron becomes visible and impacts the turbidity of the water and is typically not accepted by customers because the ferric iron within the water when in contact with surfaces, will create colloidal precipitates causing discoloring to all that it encounters over time.
Read More
Topics:
aeration,
Ferrous Iron,
Iron
Treating Hydrogen Sulfide for Environmental Safety
Read More
Topics:
odor control,
aeration,
water treatment,
advanced treatment solutions,
biological scrubber,
water plant,
odor control scrubber,
hydrogen sulfide (H2S),
Chemical Odor,
pH levels,
Decarbonation,
dissolved gases,
wastewater,
carbon dioxide,
degasifier,
gases,
Amine,
H2S Degasifier
When do you know if your decarbonation system needs service?
When a degasification tower or decarbonator becomes fouled, several indicators identify you may have a problem or that it's time to clean your system. If the efficiency of the degasifier has dropped, you will see an increased consumption rate of chemicals. If you remove less hydrogen sulfide gas from the degasifier, chlorine consumption will increase. When you increase the amount of chemical reaction occurring in the water, you will see an increase in the TSS levels and a drop in water quality. As the H2S reacts with chlorine, more solids will form and be present in the water, and the water quality will diminish.
Another indicator of a fouling condition is the pH adjustment in the Industrial Water Treatment industry. You are required to meet the set standards. As the performance of the tower drops, the removal of CO2 will also drop, leaving a higher pH level than may be desired. A quick inspection to check out the media bed should be performed. Also, do not forget to inspect the distribution system at the top of your tower and remember that all distribution systems are not alike, and inspecting the condition of each of them may require additional effort on your part. With a header lateral system, you need to inspect the distribution nozzles, but with a Weir or Tray type, you will need to check the amount of scale or fouling building up on the Weir edge or in the bottom of the pan. If the Weir edge becomes fouled unevenly, it will create "Channeling" of the water and increase the initial hydraulic load to a concentrated point on the media bed.
Read More
Topics:
water treatment issues,
blower maintenance,
aeration,
water treatment,
advanced treatment solutions,
degasifier,
Deagasification,
decarbonation of water
Do you think all distribution systems are made equal?
if you do you may be surprised that there is a lot of variation in manufacturing protocols for aerators, degasifiers, and decarbonators. Aerators are often found in use at Industrial Water Treatment and municipal water treatment facilities around the globe.
For water treatment, you may be surprised to learn that one of the key items that separate different types of aerators and decarbonators for water treatment is the type of distribution system it utilizes. To improve Carbon Dioxide (CO2) or Hydrogen Sulfide (H2S) removal you need to select the best distribution system for the tower and make sure it's maintained. Now, there are many types of aerators in general and the term is used broadly. From floating pond aerators to wastewater aerators, to vertical tower aerators, decarbonators, and degasifiers for industrial water treatment aerators. We will focus on vertical tower aerators for industrial water treatment. All types of Aerators and even degasifiers and even decarbonators and Odor Control Scrubbers require some type of distribution system to begin the process of gas transfer and to remove Hydrogen Sulfide (H2S) from water or Carbon Dioxide (CO2). It is important to evenly distribute the water or chemical solution across the media bed.
There are several types of distribution systems available and the three most common ones you will see on the marketplace are the “Tray” type, Weir, or the header lateral utilizing gas release “Nozzles”.
The selection of what type of distribution system is typically driven by the marketing side of who is selling you the tower. But in terms of real performance a distribution system utilizing a nozzle system will outperform a tray-type distributor. All packed towers are designed utilizing Henry’s Law Constant” theory of chemistry and what all towers rely upon is some type of method to break the surface tension of the water and expose the molecules of gases so that they either can escape or can be introduced to a reaction agent.
When towers are designed it is important to properly hydraulically load the top of the media bed. This is considered " Degasification Basics". This is important for many reasons and we will address these points in future updates. When using a properly designed nozzle distribution system such as a DeLoach Industries header lateral system then you get the benefit of both proper hydraulic load across the bed and you also gain anywhere from 4-10% removal efficiency depending upon the application. When looking at a chemical scrubber versus a biological scrubber you will notice they too have very different distribution systems. DeLoach Industries, Inc. has learned over its 60 years in business how to maximize gas transfer release. If designed and built properly the gas release process or interaction process (if designing a scrubber) has already begun “before” it enters the media bed.
Read More
Topics:
water treatment issues,
aeration,
Decarbonation,
De-Aeration,
decarbonator,
degasifier